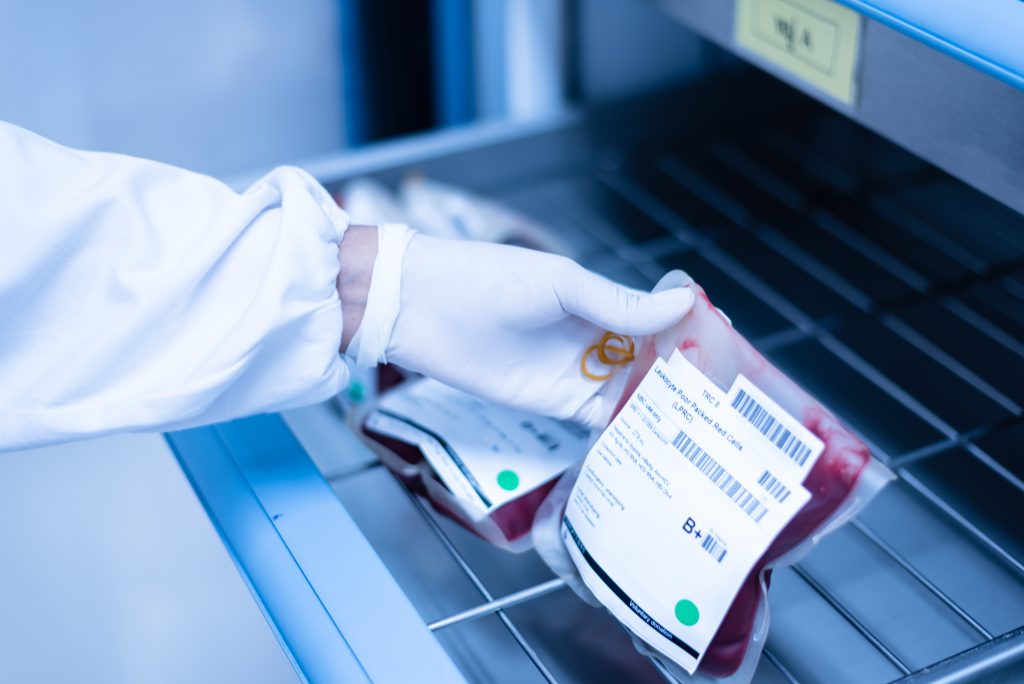
Data availability and quality problems affect 80% of pharmaceutical organizations. These data integrity gaps create compliance vulnerabilities—especially during the fragile transitions between laboratory research, manufacturing, and distribution.
To achieve true compliance, organizations must treat GxP not as separate regulations for Good Laboratory Practice (GLP), Good Manufacturing Practice (GMP), and Good Distribution Practice (GDP), but as a continuum.
Recent industry trends underscore the urgency of this approach. With only 34% of adults trusting the pharmaceutical industry, regulators are demanding more transparency and cross-functional data sharing in place of isolated logging.

The Three Pillars in Practice
To close the gaps, operational leaders must focus on three core pillars that persist regardless of the product lifecycle stage:
- Traceability: Simply archiving temperature and humidity history isn’t enough. This data needs to be contextualized and attributable across the full product lifecycle. You need the ability to link environmental conditions directly to specific batch records and ownership transfers to prove that quality was never compromised.
- Accountability: Compliance must move beyond “who signed this” to “who responded.” Detailed user activity proves a culture of compliance. This includes robust internal roles, user access controls, and audit trails as required by FDA 21 CFR Part 11 .
- Data Integrity: In an era of digital threats, integrity means ensuring records are enduring and available. FDA regulations require that closed computer systems have technological controls to protect data, ensuring records are authentic and incorruptible.
The High Cost of Data Silos
The transition from GLP (Research & Development) to GMP (Commercial Manufacturing) is often the most fragile moment in a product’s lifecycle. For large operations exceeding $1 billion in revenue, the impact is even more pronounced, with 53% of companies reporting data silos that derail the efficiency of cross-functional collaboration.
- Data Loss: When data is not continuous, it can be lost during the handoff between departments or facilities. This is critical because modern treatments are increasingly sensitive to temperature fluctuations and require tedious data collection to ensure efficacy. If this data is lost, patient safety is compromised.
- Money & Time Waste: Manual data aggregation from legacy systems introduces human error, compromises data integrity, and leads to costly rework. Upgrading to automated, cloud-based solutions can save manufacturers money by reducing manual entry errors and freeing up employees for higher-value tasks.
- Noncompliance: Disconnected systems lead to gaps in audit trails. Continuous monitoring and digital logs are essential to adhere to regulatory requirements and reduce the risk of non-compliance during audits.
How Dickson Fixes Data Silos
OCEAView Cloud, and On-Premise nvironmental monitoring systems acts as the continuity layer for your GxP operations. It is designed to meet rigorous compliance standards found in heavily regulated environments, ensuring your business has a reliable tool to safeguard assets.
Advanced Traceability
DicksonOne deploys a single cloud-based dashboard for 24/7 monitoring from anywhere—whether you are checking an incubator in a GLP lab or a warehouse in a GDP facility. This automation pushes data to a central location, bypassing the limitations of legacy systems and ensuring that your environmental data is always audit-ready.
Automated Consistency & Accountability
To solve the “who responded?” challenge, DicksonOne features programmable excursion management, allowing you to set up to 3 high and 3 low alarm limits per sensor. If an alarm is triggered, the system immediately notifies the right people via e-mail, text, or voice message, requiring acknowledgment via PIN code.
This confirms that the response to a deviation is standardized and documented, proving the accountability that regulators demand.
Scalable Integrity from Pilot to Production
As your operations grow from R&D pilots to full-scale commercial production, your compliance framework must scale with you. DicksonOne is built for enterprise growth, allowing you to manage thousands of devices and enforce Standard Operating Procedures (SOPs) centrally. With secure authentication via single sign-on (SSO) and over-the-air updates, DicksonOne ensures that your data remains secure and compliant with 21 CFR Part 11.
Whether you are upgrading legacy systems or designing a new process, we are ready to partner with you and provide assurance that your data remains secure.
We have a lot of experience helping pharmaceutical manufacturers to be fully compliant and audit-ready. Contact a Dickson expert if you want to learn more.
The Cold Hard Fact: Seasonal Changes Require Seasonal Mapping
Facility managers often feel a sense of accomplishment after their initial mapping is complete. However, that data is only a snapshot. Environmental dynamics shift significantly during extreme cold events, so your…
5 Key Questions to Verify Your Environmental Monitoring System Is FDA Compliant
In pharmaceutical manufacturing, your environmental monitoring system is your first line of defense in protecting product quality and patient safety. But simply having one in...
Audit Ready and In Control: 3 Ways to Simplify Your Blood Bank Operations
For blood banks and pathology labs, the mission is clear: to protect the lifesaving supplies that patients depend on. Your team is dedicated to this…
6 Must-Have Environmental Monitoring Features for Pharma Manufacturers
For pharmaceutical manufacturers, you know environmental monitoring systems are a key part of any quality management system, essential for pharmaceutical storage control, and serve as…
Ready to chat?
Talk To A Specialist
Our trained team of professionals is ready to help you through the compliance process from start to finish. Reach out today to start the conversation. Call +33 4 99 13 67 30 or send us a message.